 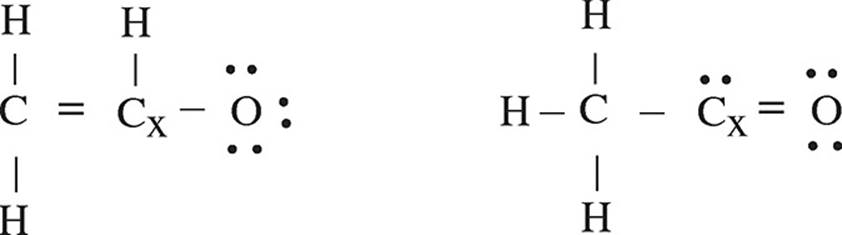
Sessions get recorded for you to access for quick revision later, just by a quick login to your account. Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts. WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. The lowest the formal charge, the more will the stability of the molecule. įinally, once the structure is drawn, it is to ensure that the Formal Charge of the molecule is lowest, as it is inversely proportional to the stability of the molecule. For example: in the case of NO 3 -, students will draw 6 dots (3 lone pairs) around each of three O atoms, keeping the N in the centre, which also makes a covalent bond with each of three O atoms, in NO 3. įollowing the octet rule of Chemistry, students will represent electrons as dots around each atom of the molecule. Start drawing a diagram, keeping that atom in the centre, which connects other atoms of the molecule, like N, in case of NO 3 - as N (Nitrogen) makes 3 bonds with O (Oxygen) in NO 3. Students will now sum up the total number of valence electrons of the molecule, which is = 5+18+1 = 24. Plus, the ionic charge present in NO 3 -, which is = 1. 
Now, we will write down the number of electrons present in each atom of the molecule (NO 3 - ). (Please note: NO 3 - is not a neutral molecule, but an ionic molecule, with a negative charge, = a.) Below are some steps that the students may consider and follow, in order to make or draw Lewis Dot Structure, these steps are:įirstly, let us take a molecule, say - NO 3 - for which we will learn to draw the Lewis Dot Structure. The diagram consists of valence electrons or electrons from the outermost shell of atoms in the form of Dots, so as to make Lewis Electron Dot Structure of atoms. Lewis dot structure is the representation of electrons of atoms, with the help of a diagram. They depict the irregular arrangement of electrons around the atom. Lewis structures are generally helpful when it comes to lighter elements and not that helpful when it comes to transition metals including both actinides and lanthanides. This can mostly be seen in higher atomic numbers. There are chances that the number of valence electrons can exceed more than 8 electrons. In some cases, the central atom can also form more than the other atoms connected to it. But, the atoms can do this and also form molecules that are not stable. Atoms in general, try and seek to half-fill or fully-fill their valence electron shell. But in reality, there are many exceptions when it comes to the structure in reality. Lewis structures are really helpful when it comes to learning about the oxidation states, valence, and the type of bonding. In general, the lowest energy structure has the smallest formal charge on the atom and also has the most distributed charges too. 
Having knowledge about the lowest energy structure can help in predicting the major product of a given reaction and also helps in understanding the phenomenon a lot. They help in selecting the lowest energy structure from all the possible Lewis structures of a given molecule. The formal charge is a theoretical charge and thus does not indicate any real charge separation present in the molecule. Thus below are the steps to draw the Lewis structure.ĭetermine the number of valence electrons present in the molecule. 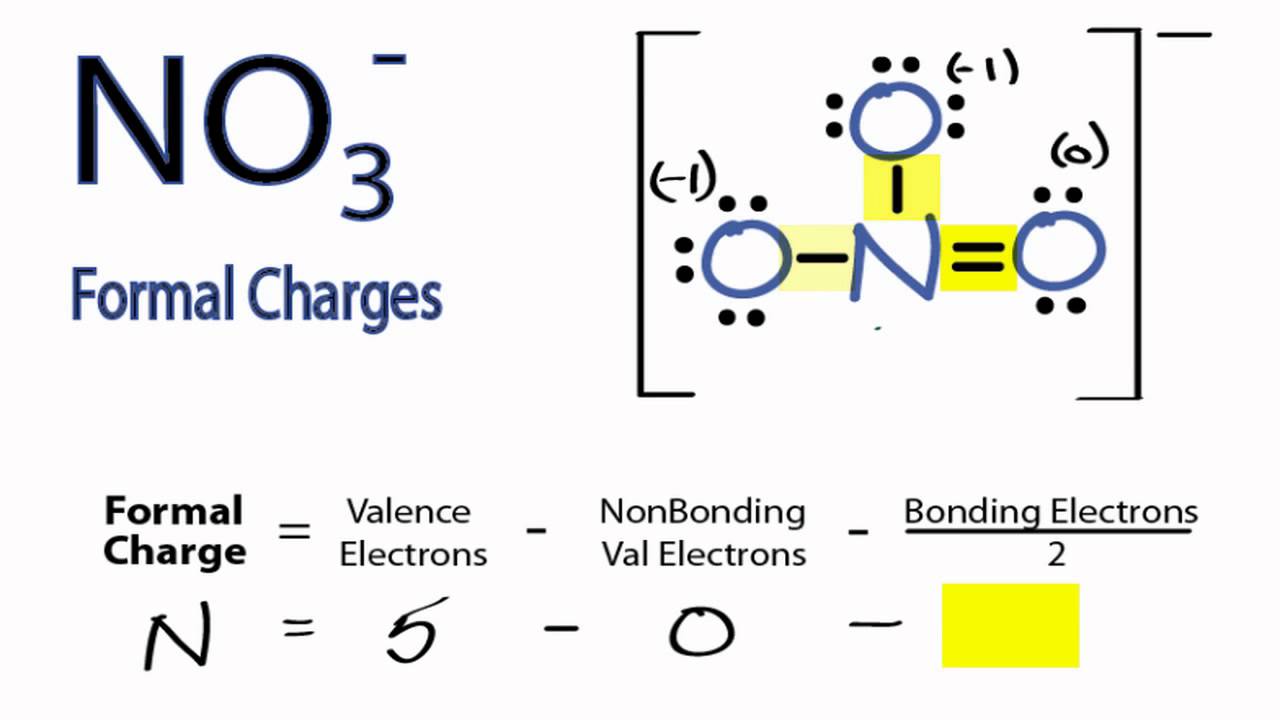
How to draw Lewis Structure: Steps with Exampleįormal Charge and its Mathematical formulaĬalculation of the Formal Charge for SO 4 -2īelow are the Steps to Find the Lewis Dot Structure.ĭrawing the Lewis structure is very important as it is then only when one can calculate the formal charge correctly. There are the following topics covered in this article: Learning to make proper Lewis dot structures can help in solving the problem that most of the students have. It also helps in predicting the geometry of the molecule. The major reason why learning Lewis dot structure is important is that it helps in predicting the number and type of bonds which can be formed around an atom. Lewis structure is basically a graphic representation of the electron distribution around an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
